DIY Genome Sequencing at Home: What It Means for Biotech

Key Takeaways

- Consumer genome sequencing hardware now costs under $5,000 for a complete setup
- AI tools like Claude are enabling non-specialists to interpret complex biological data
- The $900 per-run consumable cost makes repeated testing viable for personalized medicine applications
According to [Tom's Hardware](https://www.tomshardware.com/tech-industry/artificial-intelligence/biohacker-claims-to-have-sequenced-their-own-genome-at-the-kitchen-table-with-m3-ultra-mac-studio-claude-and-a-usd3-200-sequencer-diy-project-requires-100gb-of-data-storage-per-run-oodles-of-ram), a biohacker has successfully sequenced their own genome at home using a $3,200 Oxford Nanopore MinION sequencer, an M3 Ultra Mac Studio, and Claude AI to interpret the results. This isn't a lab stunt. It's a signal that biotech's barriers to entry are collapsing faster than most business leaders realize.
If you're running a healthcare startup, investing in biotech, or simply tracking where enterprise technology is heading, this kitchen-table experiment deserves your attention. The same democratization pattern that brought computing from mainframes to smartphones is now happening in genomics.
What Does DIY Genome Sequencing Cost Today?
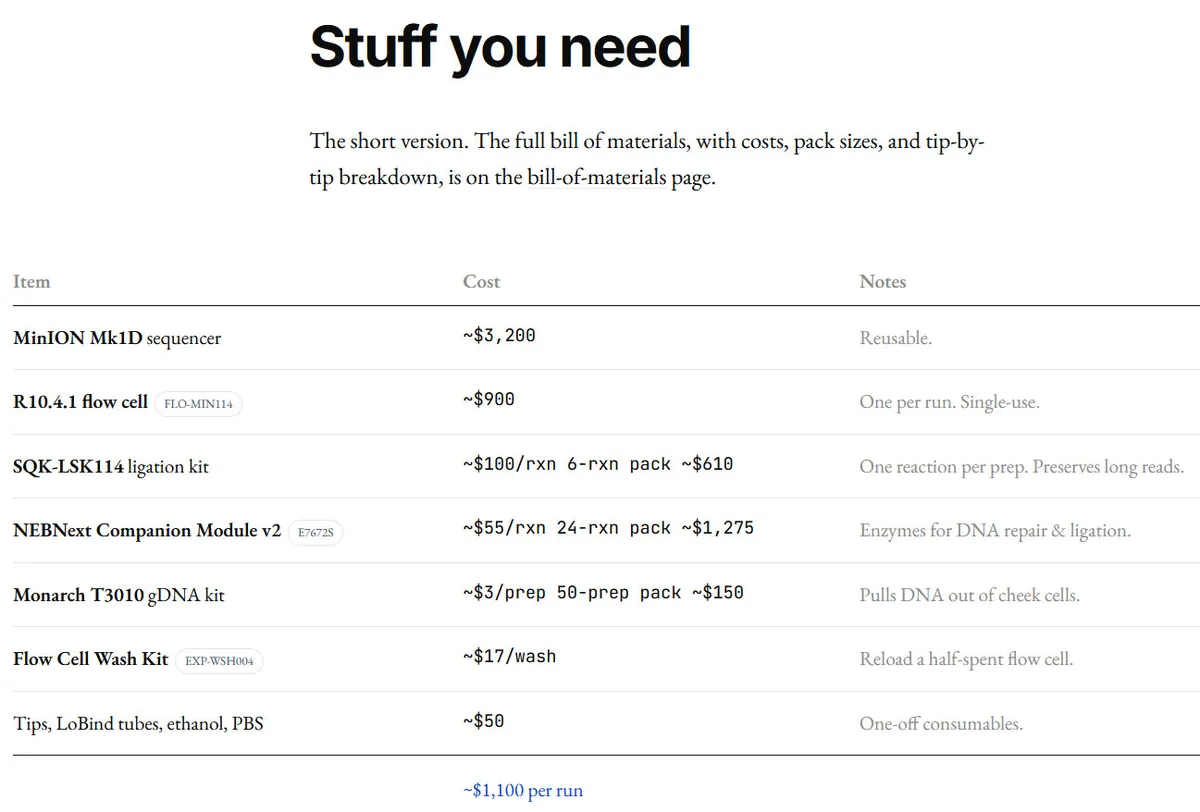
Let's talk numbers. The biohacker's bill of materials breaks down into surprisingly accessible components. The MinION Mk1D sequencer runs $3,200. It's pocket-sized, USB-powered, and capable of reading an entire human genome. The R10.4.1 flow cell costs $900 and is single-use. Every time you run a sequencing session, you burn through one.
| Component | Cost | Notes |
|---|---|---|
| Oxford Nanopore MinION Mk1D | $3,200 | One-time purchase, palm-sized |
| R10.4.1 Flow Cell | $900 | Single-use consumable per run |
| Mac with Apple Silicon (M3+) | $2,000-$8,000 | Needs 100GB+ storage per run |
| Lab consumables | $200-$500 | Extraction kits, chemicals |
| Total First Run | ~$5,000-$6,000 | Subsequent runs: ~$1,100-$1,400 |
Compare this to 2001, when sequencing a human genome cost $100 million and took years. Or even 2010, when it still ran north of $50,000. We've crossed a threshold where a motivated individual with a few thousand dollars can do what once required institutional resources.
Why Should Business Leaders Care About Home Genomics?
Three strategic shifts are happening simultaneously. First, the hardware is now consumer-accessible. Oxford Nanopore designed the MinION for field researchers, remote clinics, and yes, curious individuals. It's not a toy. It's the same technology used in COVID surveillance and cancer research.
Second, AI is filling the expertise gap. The biohacker used Claude to generate BED files. These are essentially targeting instructions that tell the sequencer which genes to examine more closely. Previously, you'd need a bioinformatics specialist. Now you need a well-crafted prompt. This pattern will repeat across every technical domain.
Third, the use case here was deeply personal. A family history of autoimmune disease that had already affected a sibling under 40. This is the future of healthcare: individuals taking proactive control of their genetic information rather than waiting for symptoms to appear.
The Compute Requirements
Each sequencing run generates approximately 100GB of data. The biohacker used an M3 Ultra Mac Studio, but noted that any recent Apple Silicon Mac with sufficient RAM works. For faster processing, an Nvidia DGX Spark could complete analysis up to 5x faster. Budget for serious storage if you're planning multiple runs.
How AI Is Enabling Non-Experts to Interpret Genetic Data
Here's where this gets interesting for anyone building AI products. The biohacker used Claude's adaptive sampling functionality to target specific genes related to autoimmune conditions. Instead of sequencing the entire genome at equal depth, they concentrated resources on the regions that mattered most.
This is a pattern we're seeing across industries. AI isn't replacing experts. It's making expert-level decision-making accessible to non-specialists. The biohacker didn't become a geneticist. They became someone who could ask the right questions and interpret the answers with AI assistance.
For business leaders, the question isn't whether this will happen in your industry. It's when. If AI can guide someone through genome interpretation, it can guide your customers through complex product configurations, regulatory compliance, or technical troubleshooting.
Major AI investments are reshaping what's possible in biotechnology
What Are the Limits of DIY Genome Sequencing?
The biohacker was careful to note this isn't a replacement for clinical testing. Kitchen-table sequencing doesn't match the accuracy or regulatory rigor of a certified lab. Error rates are higher. Quality control is nonexistent. And crucially, interpreting results correctly requires medical context that AI can't fully provide.
✅ Pros
- • Dramatically lower cost than clinical sequencing
- • Complete privacy. Your data never leaves your home
- • Ability to target specific genes of personal interest
- • Repeatable testing as technology improves
- • Educational value for understanding your own biology
❌ Cons
- • Lower accuracy than clinical-grade equipment
- • No regulatory oversight or quality certification
- • $900 consumable cost per run adds up quickly
- • Requires significant technical comfort
- • Medical interpretation still needs professional guidance
For now, this is best viewed as a complement to professional healthcare, not a replacement. Think of it like monitoring your heart rate with an Apple Watch. Useful data, but you still see a cardiologist for serious concerns.
The Personalized Medicine Market Opportunity
The global personalized medicine market is projected to reach $796 billion by 2028, growing at roughly 11% annually. Consumer genomics is a subset of this, but it's the tip of the spear. As more people understand their genetic predispositions, demand for tailored treatments, preventive interventions, and lifestyle recommendations will explode.
The business implications extend beyond healthcare. Insurance companies will grapple with genetic information in underwriting. Employers will face questions about workplace wellness programs. Pharmaceutical companies will shift toward targeted therapies. And tech companies will compete to be the platform where people store, analyze, and act on their genetic data.
Trust issues with AI-interpreted data affect genomics too
What This Means for Healthcare and Biotech Startups
If you're building in healthcare or biotech, three opportunities emerge from this trend. First, there's a clear gap in the interpretation layer. Raw genetic data is useless without context. Startups that can bridge genetic information and actionable health recommendations will capture significant value.
Second, the data storage and privacy challenge is massive. Each run generates 100GB. People who do multiple runs over years will accumulate terabytes of sensitive biological information. Secure, private, accessible storage is a product waiting to be built.
Third, consider the education market. The biohacker learned genomics well enough to run this experiment through self-study and AI assistance. There's demand for structured learning paths that combine these tools with domain expertise.
Oxford Nanopore's MinION explainer video above shows just how accessible this technology has become. The device fits in your palm and connects via USB. That simplicity is the point.
The Hardware Arms Race in Consumer Biotech
The biohacker noted that while an M3 Ultra Mac Studio works well, an Nvidia DGX Spark could process results up to 5x faster. This hints at a coming hardware differentiation in the consumer biotech space. As sequencing becomes more common, compute performance becomes the bottleneck.
For tech leaders, this is familiar territory. We've seen this pattern in gaming, video editing, and AI inference. Early adopters make do with available hardware. As the use case matures, specialized silicon emerges. The question is whether Apple, Nvidia, or a startup will own the consumer bioinformatics compute layer.
Energy infrastructure investments show where compute-heavy industries are heading
Regulatory and Ethical Questions Ahead
This technology will force regulatory conversations. What happens when someone sequences their genome, finds a concerning variant, and their insurance company asks about pre-existing genetic conditions? How do employers handle genetic information in wellness programs? What are the liability implications for AI systems that help interpret medical data?
For now, the regulatory framework treats this as a gray area. DIY sequencing for personal research isn't explicitly regulated in most jurisdictions. But as adoption grows, that will change. Companies building in this space should anticipate compliance requirements and design for them early.
Frequently Asked Questions
How much does DIY genome sequencing actually cost?
Initial setup runs about $5,000-$6,000 including the MinION sequencer ($3,200), flow cell ($900), a capable computer ($2,000+), and consumables. Each subsequent run costs approximately $1,100-$1,400, primarily due to the single-use flow cell.
Is DIY genome sequencing accurate enough for medical decisions?
No. The biohacker explicitly noted that kitchen sequencing doesn't match clinical accuracy or rigor. It's useful for education and preliminary research, but medical decisions should still involve certified labs and healthcare professionals.
What computer specs do I need for genome sequencing?
You need at least 100GB of free storage per run and substantial RAM. The biohacker used an M3 Ultra Mac Studio but noted any recent Apple Silicon Mac (M3 or later) works. Nvidia DGX Spark can process results up to 5x faster.
What's the business opportunity in consumer genomics?
Three main areas: interpretation layers that translate raw data into actionable insights, secure storage solutions for terabytes of sensitive genetic data, and educational platforms that combine sequencing tools with domain expertise.
How does AI help with genome sequencing?
AI tools like Claude can generate targeting files (BED files) that direct the sequencer to focus on specific genes of interest. This replaces the need for a bioinformatics specialist and makes the technology accessible to non-experts.
Logicity's Take
At Logicity, we build AI agents and automation systems for businesses. While we're not biotechnologists, this story resonates with patterns we see daily. The biohacker used Claude to generate BED files. That's essentially using an AI agent to bridge a knowledge gap. This is exactly what we build for our clients: AI systems that help non-specialists accomplish expert-level tasks. For Indian tech businesses watching this space, the opportunity isn't in building sequencers. It's in the software layer. Interpretation dashboards. Secure data storage. AI assistants that explain results in plain language. These are software problems, not biotech problems. The compute requirements (100GB per run, serious RAM needs) also signal infrastructure opportunities. Indian cloud providers and data center operators should note that biotech workloads are coming to consumer markets. The same pattern that drove cloud gaming and AI inference demand is about to hit personalized medicine. We're also watching the trust angle carefully. Our work on AI systems has taught us that users need confidence in AI-generated insights before acting on them. In genomics, the stakes are higher than in most domains. Whoever solves the trust problem in AI-interpreted medical data will own this market.
Need Help Implementing This?
Logicity helps businesses build AI-powered tools and automation systems. If you're exploring how AI agents can bridge expertise gaps in your industry, whether that's healthcare, finance, or technical services, we'd love to talk. Our team specializes in Claude-based agents, workflow automation, and enterprise AI integration. Reach out to explore what's possible for your organization.
Source: Latest from Tom's Hardware
Huma Shazia
Senior AI & Tech Writer